Koen
25 m / sec is the minimum air speed at low RPM would be desirable to have ?
Dumb question!!! When you folks say 25 m/sec, is that like sucking all the air out of a box 10ft x 10ft x 7ft. in one second??TomC
l havent measured it but l know its white hot becouse it shines like a light bulb out the nozzle. White light apears above 2000 C. [quote=“k_vanlooken, post:40, topic:2453”]
I doubt you have a higher temperature then 1300°C to 1600°C coz all ashes would be vitrified and the metal molten…
[/quote]
l have to desagree. The ash does melt and flow down and around the tube like molten glass so there is a lot more thain 1300 C. The metal cant melt becouse of its mass.
The exterior gets warm when l come home noting critical.
The gas exits trugh the wire mesh on top.
l do not use any cooler for the gas. l only drive about 30km to my job so the gas barely gets warm at the time.
TomC,
In Miles per Hour, it works out to 55.9234. Or 90 Kilometers/hour. You might be thinking about cubic meters per second?
Koen,
I wish that I understood your analysis and the chart I inserted. Why are you using 4000 RPM in the calculation? Doesn’t CO reach its maximum power around 2500 RPM when the piston speed starts to exceed the CO flame speed? You can probably tell from my question that I don’t understand. Would three holes give greater airspeed and higher quality gas?
Bottom line: If I were to try Kristijan’s nozzle set up on my 1800CC MGB, what in your opinion would be a good size to start? Would 4, 10mm holes like his give me greater airspeed and better gas? If I used a slot instead of holes, what opening area would be optimum?
Kristijan,
I went to the shop and put slice and a plug in my 5/8 inch ID X 1’1/8 OD copper nozzle.
I had good gas in 30 seconds-wow. At 12 minutes something opened up and started burning the outside of my barrel. Who wants to guess what happened? 1) the small piece of steel pipe that i used to plug the end, burned through and fell off? or 2) the brass end cap melted or burned off? or 3) the copper pipe opened up and the steel pipe fell out? or 4) other? I’ll inspect tomorrow and give the results.
Thank you Ray. I can go back to bed now. They say you should learn something new every day and that is new to me. In all my years I have never heard of “air blast velocity”. Good nightTomC
Hi Bruce,
RPM: a generator set on a honda clone runs at 3600 Rpm for 60 Hz
CO flame speed: flame speed changes under pressure but still would be limited above the 3800 Rpm marker, the gas would still be burning when the exhaust valve opens
Most gas from charcoal gasifiers have a % H2, that speeds up the flamespeed a lot… and make it more useable at higher rpm
I use the same charts as reference , to make good gas, my advice: use those value’s
for your setup:
For the original spreadsheet i will post a link later , so this would be available for use
The spreadsheet / variables to be altered, will be in yellow, outcome in green ? 
I work with the multiple nozzles since 2 years now, even with automatic open and closing from nozzle openings… only pro’s not con’s so far.
I am very positive about this work from Kristijan, now only to give him some corrected background/research to use the correct terminology 
Hi Tom,
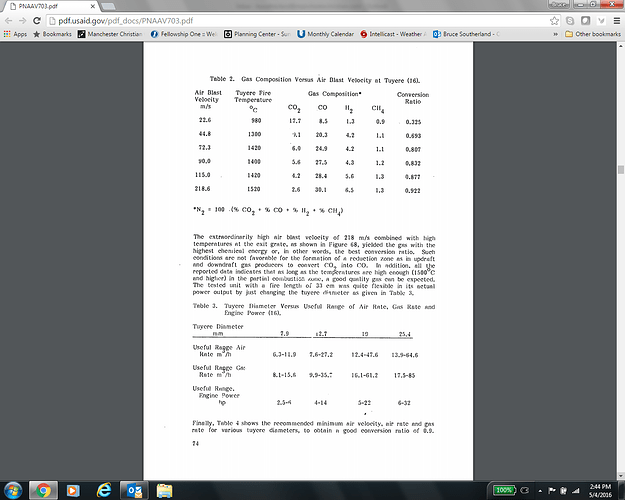
This chart is very informative, its perfect research data ready for use…
This link will lead to the excell sheet
https://drive.google.com/open?id=0B4CL3bqit0osdHU3OVMyeE1yeHM
if you want to obtain an desired airspeed at the nozzle opening as per charts earlier published and or want to use more then 1 nozzle,… fill in the fields and the results will be shown…
To remind, this are speeds at wot and in full efficiency of 0,7, real values are less at lower rpm’s, but that you can change also
Hi Kristijan,
1: i admire your work , so please see my reply’s as a positive critic only, nothing as negative…
The next picture show’s the temperature we observe:
And this link gives the melting temperatures from different materials
http://www.engineeringtoolbox.com/melting-temperature-metals-d_860.html
Same value’s here:
And this paper contains a lot of information of ash behavior and metal oxidation…
http://www.diva-portal.org/smash/get/diva2:426924/FULLTEXT01.pdf
To all,
I forgot to mention 
1: its hot inside the glowing charcoal
2: any gas will do but good gas will do better
3: the limits are given by your own imagination… and the time you want to spent to find out where your imagination leads you…
4: and that all my work have helped you to discover some problems you didn’t knew existed 
5: There are no problems to solve, only solutions to find…
Somehow i have the feeling that i need a coffee … reading at DOW since 4 am…
Hi Tom,
25 meters per second would be:
25 cbm passing thru a tube with a cross section of 1 sqm in 1 second time…
or 25 cubic inch passing thru a tube with a cross section of 1 square inch in 1 second would be the speed of 25" per sec
if reducing the cross section to 1/4 square inch, the same amount of volume , in the same time frame, would increase the speed equal the decreasing ratio from the cross section
so , in that case, the 25 cubic inch would have a speed of 100" per second…
I would have guessed 3. Very surprised that the cap blew open. Doesn’t make sense to me.
Bill,
I was hoping that this is what i would find. My experience with copper has shown very good stability at high temps because it rapidly conducts heat away from the hot area to the outside of the reactor where it releases it to the outside air. That is why it is the metal of choice for smelting. The relatively thin and relatively poor conducting steel was no match for this blast temperature.
Koen,
Thank you very much for this good quick answer.
Blessings!
Thanks Koen. I got it now. In m / sec I didn’t recognize you were talking about velocity. If it had been ft / sec or mi / hr I would have grasp it better. TomC
I wuld go with 1) then 2). Looks like brass culdnt conduct the heat trugh iron and it just melted. Were you able to light it thrugh the cut nozzle?
Koen there realy was nothing personal in my post  l was thod white light aperas above 2000 not 1500 C based on an old book so l based my observations on that. I never realy gave temperature much atention after l started to go with stright air so that was just a guess but thanks for the correction. That is exactly the purpose of this forum-to help others fix there mistakes and others to help correct yours.
l was thod white light aperas above 2000 not 1500 C based on an old book so l based my observations on that. I never realy gave temperature much atention after l started to go with stright air so that was just a guess but thanks for the correction. That is exactly the purpose of this forum-to help others fix there mistakes and others to help correct yours.
I allsow fownd an interastyng fact that shows why the gasifier tends to give better gas when just air is injected (no exhaust). at high temperatures charcoal tends to oxidise stright to CO not CO2 and then reduce to CO. That might alsow be the reason why the generator gives good gas so fast.
The reason why you can see the difference lay’s in the amount of oxygen injected.
- oxy = High temp, = always burned C to Co2 = Exothermique reaction.
Your observation is correct about higher temperature with C = endothermique reaction , thermal destruction of C to CO. Be it not an oxydising reaction dough, it also works in the total absence from oxygen…
(similar as gasifying with superheated steam)
Also: Boudouard reaction likes higher temperatures
Less Co2 output = less consumption of charcoal for the same amount of energy output.
And you’r right about learning from each other.