Hi All,
I see some entries that have various water drip systems. I presume
the builders believe they are splitting H2O into H and O atoms and
getting more bang for their buck.
Here’s some info I found and I’ll be darned if I can figure out how a gasifier
can achieve these temps without melting the gasifier!
What am I missing?
The charcoal acts a bit like a catalyst helping to pull the bits apart. I might still have a book that studied town gas and got into the details however at this time I have a problem getting to it.
The water gas shift is an endothermic reaction.
Part of the reason we add water is to cool the reaction zone down to an optimal level.
If we add hotter steam we can use more and create more H2 and free oxygen for richer gas.
Theoretically it would be nice to put dry steam in at 400F through a separate injector to minimize excessive cooling the reaction zone or heating and thinning out the fresh air
Hi Pepe,
The benefits/secrets of glowing carbon 
"Production
Synthesis gas is made by passing steam over a red-hot carbon fuel such as coke:
H2O+ C → H2+ CO
The reaction is endothermic, so the fuel must be continually re-heated to keep the reaction going. In order to do this, an air stream, which alternates with the vapor stream, is introduced for the combustion of carbon to take place.
Theoretically, to make 6 L of water gas, 5 L of air is required.
Or, alternatively, to prevent contamination with nitrogen, energy can be provided by using pure oxygen to burn carbon into carbon monoxide.
O2 + 2 C → 2 CO
In this case 1 L of oxygen will create 5.3 L of pure water gas."
source: wikipedia
Thank you, Koen, it’s beginning to make some sense now.
Hi Pepe,
the soon as my computer gets back from repair , i will upload a clip where i show the H2 % increasing with adding water in the glowing carbon.
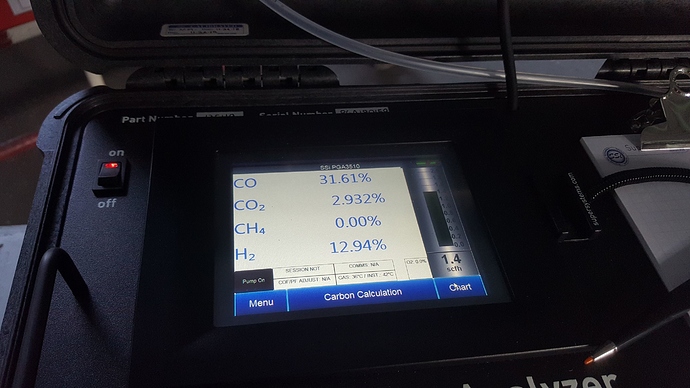
Meanwhile, here is a picture from my thread with the gas analyser
testing with water drip
Koen, Isn’t it great to have those kinds of tools!!! That is so cool!
Funny you guys should mention it, I have been thinking along just those lines myself. I have been kicking around some ideas with Kristijan about water injection, especially after catching up on his old seat arosa thread. Here is my latest thought:

Im thinking of using 1/4" copper pipe. On the last run thermocouple #1 was between 970 and 1400F (520-760C).
I think this might help keep my gas temp down, and I also suspect that having some hydrogen would really up the gas quality. Any thoughts?
Oh yeah, and I also want one of those gas-analyzers, but “If you have to ask…” ![]()
If you can get the steam up to twice the boiling point under manageable pressure of about 40 PSI or less that would be both safe and manageable.
I think the best place to do this is off the exhaust of the engine.
This way steam production is somewhat regulated by the amount of load on the engine if you see where I am going.
You could try and super heat the steam produced in a coil off the gasifire but I don’t know how much heat and pressure this could make.
The flow and pressure would depend on the jet size you chose for the injector.
Lots of experimentation required.
Yeah feed from your condensate tanks to be self sustaining. Im going to play with this as well. Might do steam direct injection into the engine along with a lower charcoal reactor steam injection system. No more daily ash removal. Goal will be to get to a two week or longer char ash servicing interval.
Not so sure thats much help Matt.
Wood has more moister locked up in the cellulose than we need.
Even bone dry wood has more than enough moister to fully convert in the water gas shift.
This is precisely why you need the condensate tank in the first place
Now if you could increase the temperature and pressure at the same time in the reduction zone ( operate above atmospheric pressure ) then I bet you could add more water/steam.
I am still convinced an exhaust driven turbo could raise the pressure and heat inside a conventional down draft gasifier to do these sorts of things, but I ain’t got a big enough engine or turbo or the welding skills to try ( you DO !! )
Doug Williams eluded to an issue with some tropical hard woods being so dense and hard they burned like coal and these did need some extra water to control the temperature.
What I mean is for the water supply for steam injection. My thinking is different because Im thinking in terms of wood gasification vs char coal. So in my system I would not be injecting the wood gasifier, I would use any moisture it created to feed the charcoal unit in its lower to control its heat.
The problem for this concept; is the new pellet reactors have a very near 100% water shift. I showed the guys at Argos last year, what it dropped after running all day. Only a table spoon of condensate after something like 8 hours running non stop. Core temps are beyond steel melting point in this reactor. How I am getting away with this is because when these temps are reached is after the bottom of my restriction. Its just open at this point no metals come in contact with the gas at this temp. From here we start the endothermic reactions and the gas is much cooler by the time it reaches the grate.
While Im on this subject. my gas exit temps are much higher. The reason this is note worthy, is because dry gas is much much harder to cool than gas full of moisture. 
Some things i can not post on a public forum ( due to big company’s seeking for profitable know how ) however i can show/teach/share them when we meet in Argos…
Picture yourselve being a person with a big imagination, then find a person that trusts your knowhow and provide you with tools to pursue your dreams/idea’s
Think what can be possible… and i am not talking about free energy… but just plain old knowhow combined with new technology’s
I am privileged coz i have DOW, the drive and some education about the old knowhow + i am that person with the imagination mentioned above…
Its not what i think that is right… its what has been done by people with different opinions that will lead to the solution needed
Most of my work is based on the words from SU: “blowing smoke thru a pile of glowing carbon”
Finding out how much smoke i can blow thru how much glowing carbon is the path i have chosen…
Try to find a way to use the heat of exhaust pipe to superheat water into steam, then pipe that into your reduction zone…
( i am there and i am doing that , just i don’t show everything and certainly not my failure’s on the way…  )
)
No we dont have failures.  This is easy!!
This is easy!!
Matt I have been trying to understand this.
Why would the moister content make that much of a difference?
Is this one of those states of matter energy transfer things?
Going from a gas to a liquid sheds heat?
Koen ,Wallace, @pepe2000,
One great thing about the folks on the DOW forum, they fire up my curiosity and cause me to research and learn new things continually. You can teach an old dog new tricks, if the dog is willing! I followed links which added up to more links. Read a bit about Fischer-Tropsch and Thaddeus S.C. Lowe. Learned about Hydrogen, and superheated steam. Hot carbon and coke. Ammonia and methane. Early refrigeration. Steam reforming. Thanks to all! 
Yeah principle of AC.
If the gas has nothing to give it stays hot.